Cancer and other Diseases
We seek to understand the tissue-level regulatory mechanisms that drive cancer and other diseases. More and more evidence suggests that acquired cancer cell mutations cannot explain alone the behavior of many malignant invasive tumors. For example, it has been shown how early embryo cells transplanted into ectopic places generate teratocarcinomas; surprisingly, when teratocarcinoma cells are injected into early embryos, they generate normal tissues. Moreover, transplanting tumor cells into a regenerating tissue, such as an amputated salamander limb, get perfectly integrated into the host tissue. These findings point toward a tissue-level explanation of cancer formation. We investigate the tissue-level interactions and regulations responsible for cancer suppression and progression. A computational approach using novel spatial models and theory of carcinogenesis at a multicellular level may be the key tool for unraveling this complex process and its underlying control mechanisms.
Publications
Discovery of dynamic models for AML disease progression from longitudinal multi-modal clinical data using explainable machine learning
R. Mousavi, M.K. Mustafa Ali, D. Lobo
medRxiv doi:10.1101/2025.04.07.25325267, 2025.
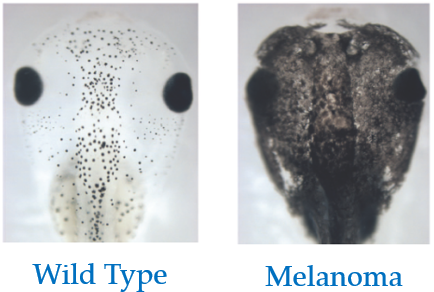
Discovering novel phenotypes with automatically inferred dynamic models: a partial melanocyte conversion in Xenopus
D. Lobo, M. Lobikin, M. Levin
Scientific Reports 7, 41339, 2017.
Serotonergic regulation of melanocyte conversion: A bioelectrically regulated network for stochastic all-or-none hyperpigmentation
M. Lobikin, D. Lobo, D.J. Blackiston, C.J. Martyniuk, E. Tkachenko, M. Levin
Science Signaling 8(397), pp. ra99, 2015.
(Reviewed in a focus paper)
Resting potential, oncogene-induced tumorigenesis, and metastasis: the bioelectric basis of cancer in vivo
M. Lobikin, B. Chernet, D. Lobo, M. Levin
Physical Biology 9(6): 065002, 2012.
(Selected for the journal cover)